PRODUCTS
Basic zinc carbonate
English name: Zinc carbonate basic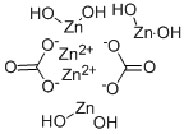
Molecular formula: 3Zn (OH) 2 · 2ZnCO3
Molecular weight: 549.1
Production method: Sulfuric acid method
Technical indicators: the main content (in terms of Zn) (dry basis) mass fraction /% ≥ 57.5%
Loss of ignition: 25.0%~28.0%
Moisture ≤ 2.5%
Sulfate (in terms of SO4) mass fraction ≤ 0.6%
Cadmium (Cd) mass fraction ≤ 0.02%
Lead (Pb) mass fraction ≤0.03%
Physicochemical properties: white fine amorphous powder.
Odorless, tasteless. The relative density of 4.42 ~ 4.45.
Insoluble in water and ethanol, slightly soluble in ammonia, soluble in dilute acid and sodium hydroxide.
With 30% hydrogen peroxide action, the release of carbon dioxide, the formation of oxides.
Main use: can be used for the manufacture of catalytic desulfurization agent, latex film products, pigments, rayon, medicine with basic zinc carbonate to do light astringent, preservatives, talcum powder, disinfectant and skin protection agent. Since the transparency of the basic zinc carbonate is close to that of the natural rubber, it is used as a reinforcing agent and a vulcanization accelerator in a transparent rubber product.
Storage: Store in a cool, ventilated, dry store. Do not mix with acid and alkali items. Pay attention to moisture. Transport process to prevent rain, damp, anti-sun, heat. Fire can be used when fighting water, sand and fire extinguishers.

Sales Department: 0086-533-7877022
Supply Department: 0086-533-7877029 / 0086-533-7877027
Office: 0086-533-7877100 Fax: 0086-533-7877022
E-mail: aethervip@126.com

- Follow us -